Aficamten achieves dual-endpoint success in pivotal non-obstructive HCM trial
TL;DR Summary
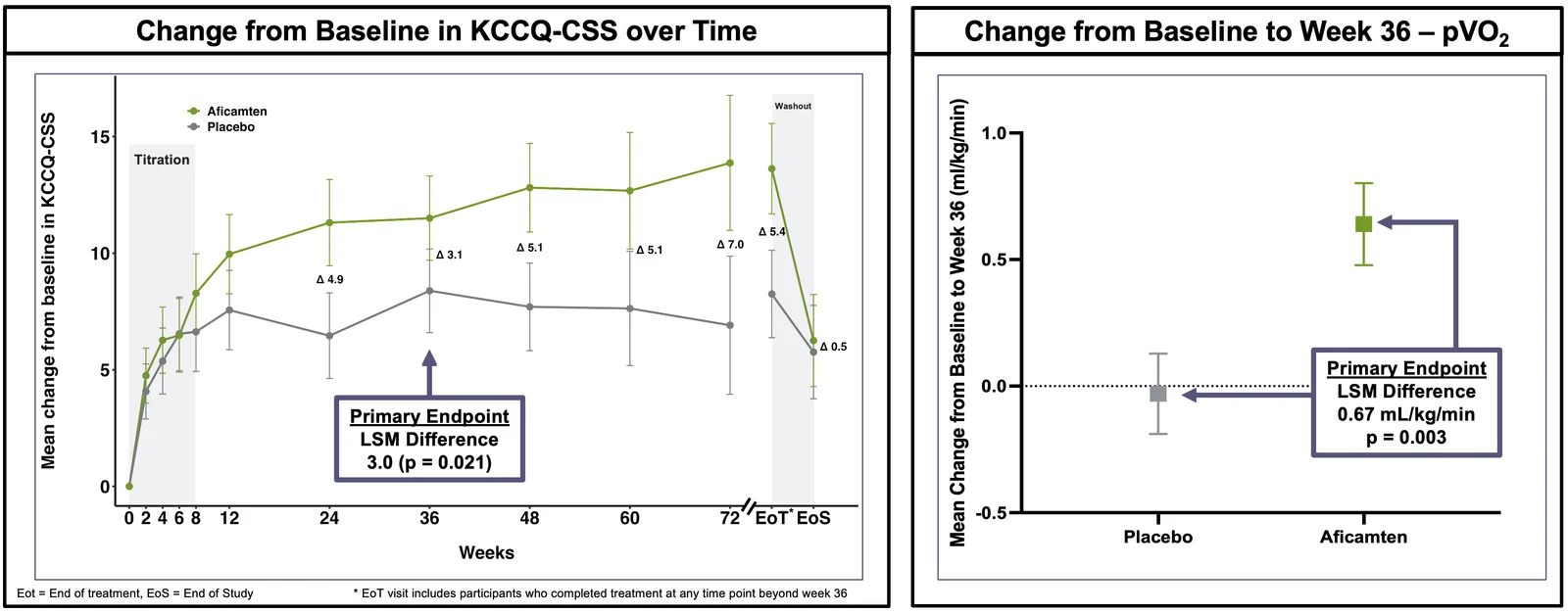
Cytokinetics announced positive topline results from ACACIA-HCM, a pivotal Phase 3 trial of aficamten in symptomatic non-obstructive hypertrophic cardiomyopathy, meeting both primary endpoints (KCCQ-CSS and maximal exercise capacity pVO2) at Week 36 with statistical significance (KCCQ-CSS 11.4 vs 8.4, p=0.021; pVO2 0.64 vs -0.03, p=0.003). The findings were robust and consistent across key secondary endpoints, and the company will host a conference call on May 5 at 8:00 AM ET. Notably, there are no approved therapies for non-obstructive HCM, and aficamten is a cardiac myosin inhibitor in development, with MYQORZO approved for obstructive HCM.
Topics:business#acacia-hcm#aficamten#healthcare#kccq-css#non-obstructive-hypertrophic-cardiomyopathy#pvo2
- Cytokinetics Announces Positive Topline Results from ACACIA-HCM, the Pivotal Phase 3 Clinical Trial of Aficamten in Patients with Non-Obstructive Hypertrophic Cardiomyopathy Cytokinetics
- Cytokinetics drug Myqorzo meets twin efficacy goals in study of genetic heart disease statnews.com
- Cytokinetics' Myqorzo succeeds in landmark cardiomyopathy trial Fierce Pharma
- Cytokinetics succeeds where Bristol Myers failed in heart muscle disease Endpoints News
- Cytokinetics to Announce Topline Results from ACACIA-HCM, the Pivotal Phase 3 Clinical Trial of Aficamten in Patients with Non-Obstructive Hypertrophic Cardiomyopathy, on May 5, 2026 Yahoo Finance
Reading Insights
Total Reads
0
Unique Readers
6
Time Saved
12 min
vs 13 min read
Condensed
96%
2,455 → 94 words
Want the full story? Read the original article
Read on Cytokinetics