FDA backs first gene therapy to treat a rare genetic deafness (Otarmeni) with free US access
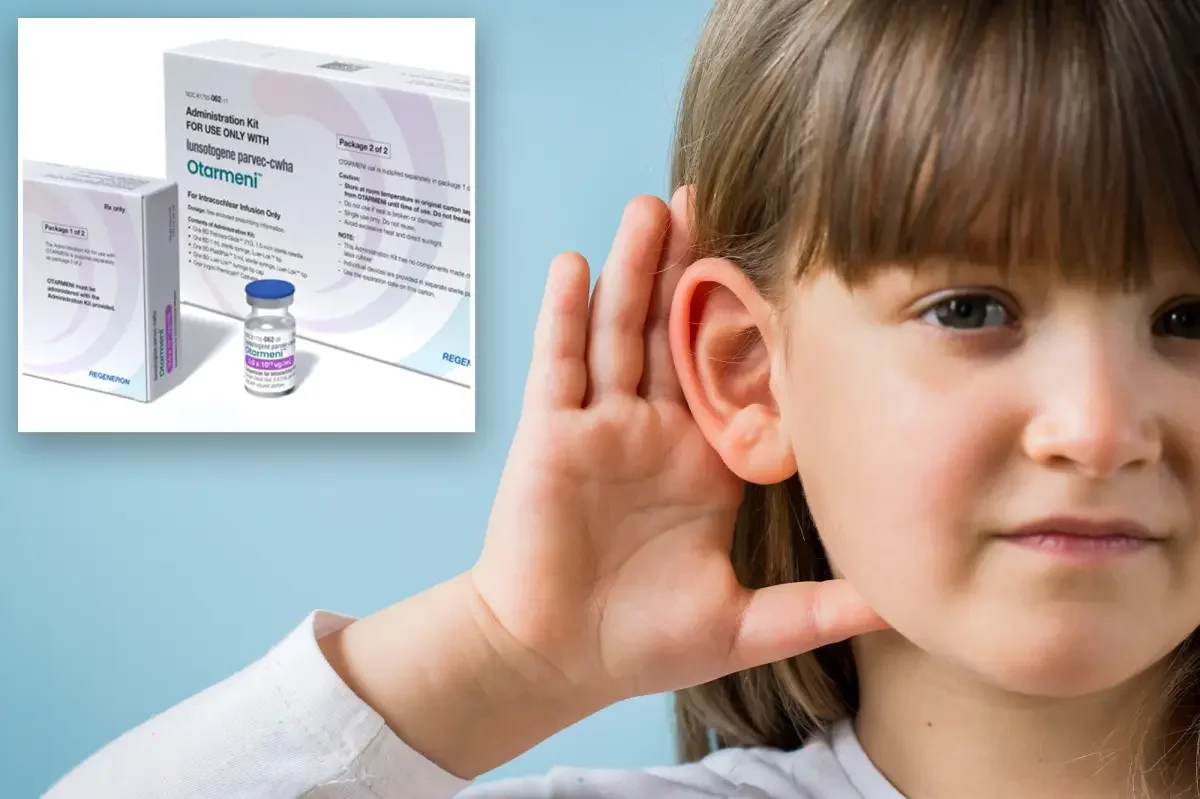
The FDA approved Otarmeni, Regeneron’s gene therapy for OTOF-related deafness—the first gene therapy for this rare inherited hearing loss—delivered via a single cochlear injection to restore otoferlin production. In a 20-child trial, several participants showed hearing improvements, with five of 12 followed for 11 months reaching near-normal hearing. Regeneron will provide Otarmeni at no cost to US patients, and the therapy was fast-tracked under the Commissioner’s National Priority Voucher program. While promising, effects were generally safe, the condition affects about 50 US babies born each year, and this advance could pave the way for more gene therapies and potentially reduce reliance on cochlear implants.
- FDA OKs ‘life-changing’ gene therapy for hearing loss — and it’s free New York Post
- New Gene Therapy Enables Children With a Rare Form of Deafness to Hear The New York Times
- FDA Approves First-Ever Gene Therapy to Restore Hearing WSJ
- Regeneron ushers in new genetic medicine era with groundbreaking gene therapy approval Fierce Pharma
- Multicentre gene therapy for OTOF -related deafness followed up to 2.5 years Nature
Reading Insights
1
5
24 min
vs 25 min read
98%
4,810 → 104 words
Want the full story? Read the original article
Read on New York Post