FDA Clears Higher-Dose Wegovy to Boost Weight Loss
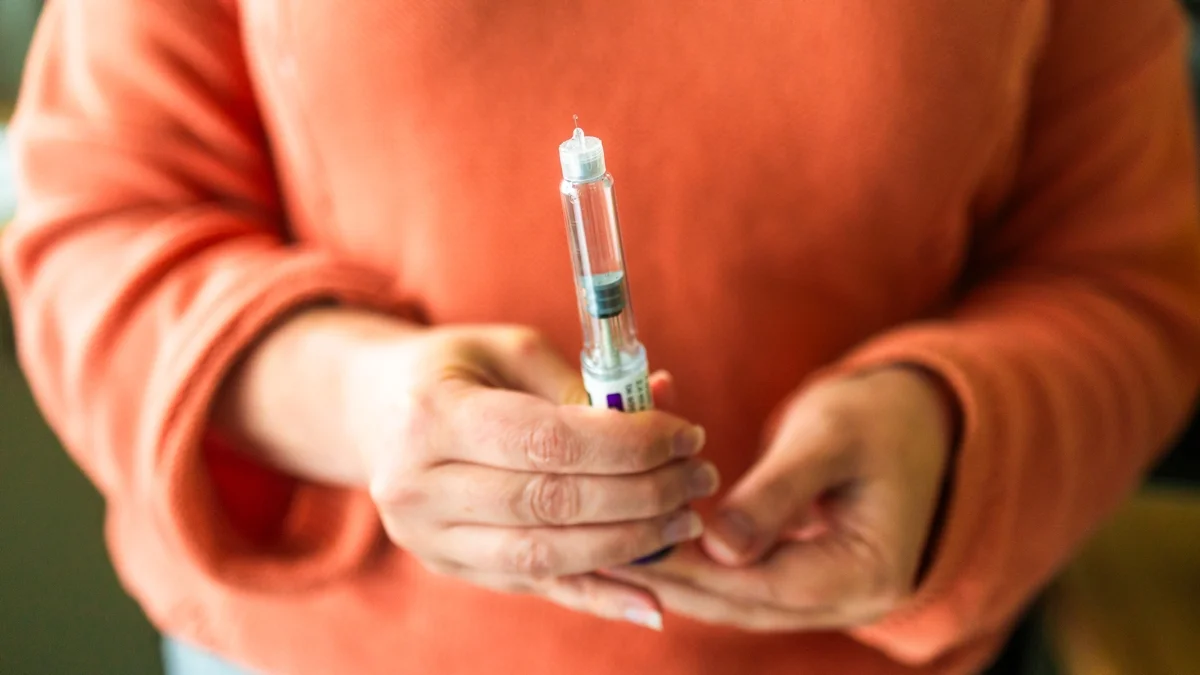
The FDA approved Wegovy HD, a 7.2 mg weekly dose of semaglutide, a higher-dose version expected to boost weight loss. In trials, the higher dose yielded about 19% body-weight loss (roughly 47 lb) vs 16% (about 39 lb) with the 2.4 mg dose over about 17 months; the drug will be available in April with price to be announced. European regulators had approved it earlier. Side effects were common (GI issues in 70%+; 23% reported skin sensations) and serious adverse events occurred in ~7% of the higher-dose group. The jump from 2.4 mg to 7.2 mg is substantial, and real-world safety will need monitoring. An oral Wegovy pill was approved by the FDA in December for context.
- FDA Approves New High-Dose Wegovy in US For Boosted Weight Loss ScienceAlert
- Weight-loss treatment is on the verge of a dramatic shift – again CNN
- FDA approves higher dose version of weight loss drug Wegovy as Novo Nordisk tries to win back market share CNBC
- Battle for weight-loss supremacy shifts from jabs to pills Financial Times
- A high dose of Wegovy that helped people lose 21% of their weight in a clinical trial was just approved MarketWatch
Reading Insights
0
44
3 min
vs 4 min read
83%
696 → 117 words
Want the full story? Read the original article
Read on ScienceAlert