Pro-Life Coalition Fractures With Trump Over Abortion Pill Access
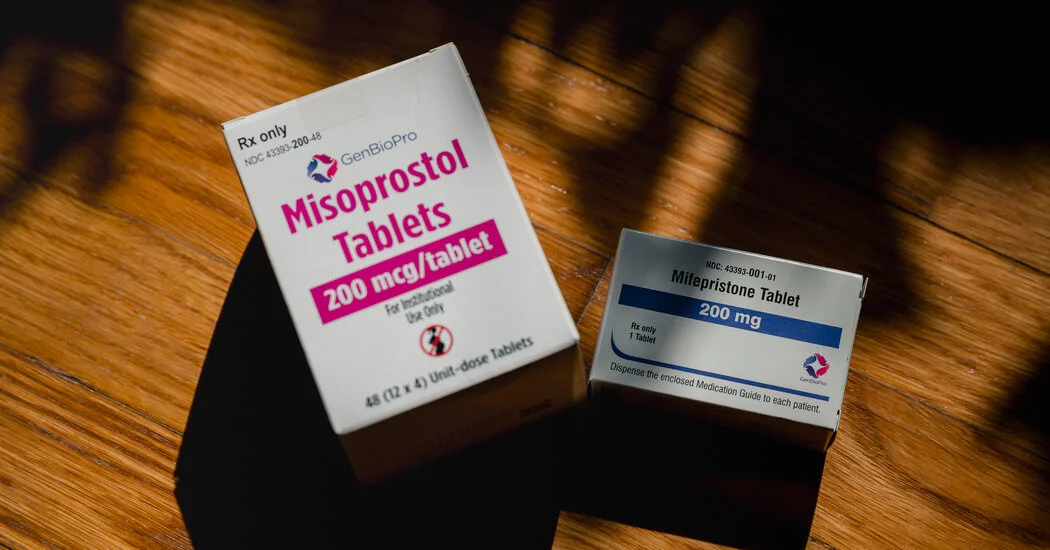
After the Supreme Court preserved access to telemedicine abortions, anti-abortion groups intensified pressure on the Trump administration to act on abortion pills and funding for Planned Parenthood, pressing meetings with the FDA and DOJ and signaling that failure could hurt GOP turnout in the midterms; they want swift use of powers to curb access, including potential moves like the Comstock Act to ban mail-order abortions and defund Planned Parenthood, though some insiders caution the White House ultimately holds the leverage.