Novo Nordisk rolls out Wegovy subscription to lock in predictable GLP-1 prices
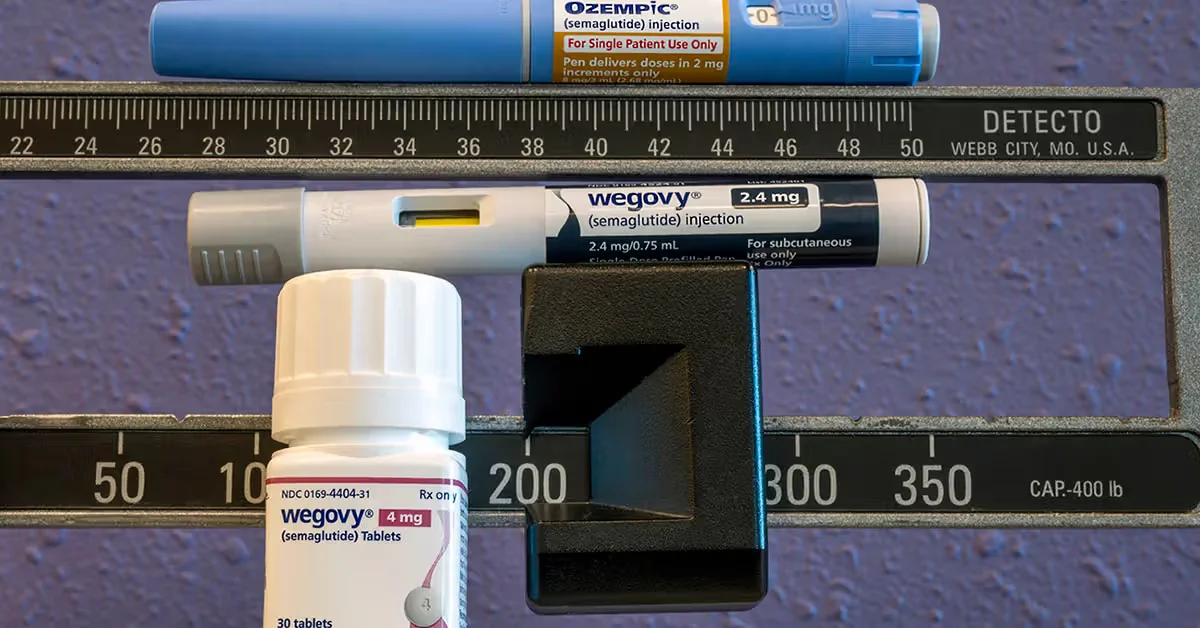
Novo Nordisk launched a multi-month Wegovy subscription for its injectable doses and the high-dose pill, offering 3-, 6-, and 12-month plans with fixed monthly prices and potential savings up to $1,200/year for the injection and $600/year for the pill, via telehealth partners; the program aims to improve adherence and pre-empt competition from Eli Lilly’s upcoming oral GLP-1, and patients can opt out anytime (not yet on NovoCare DTC).