CDC Recommends Second COVID Shot for Seniors
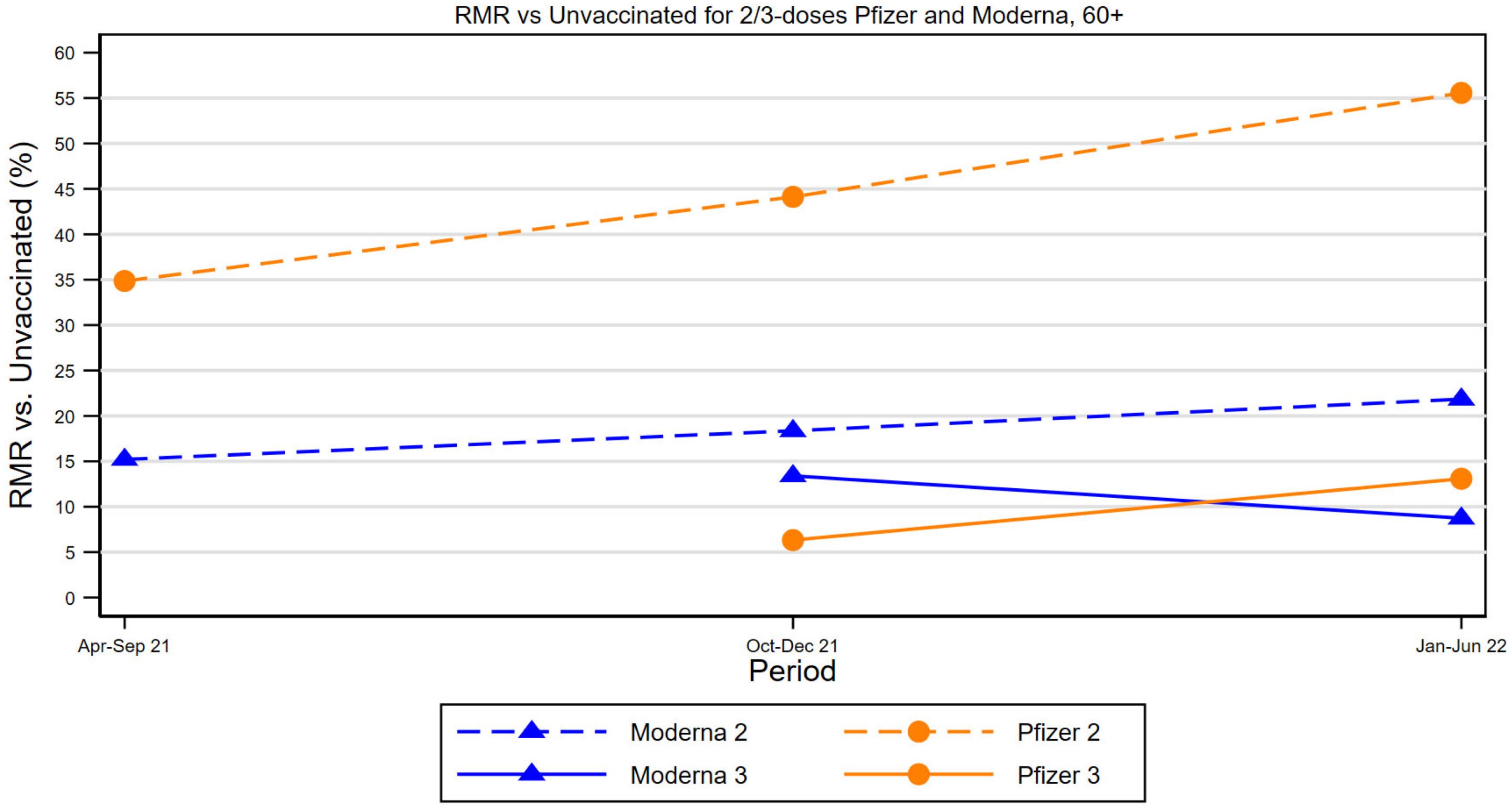
The CDC recommends an additional dose of the current COVID-19 vaccine for people aged 65 and older, emphasizing the importance of vaccination due to waning protection and the vulnerability of older adults to severe COVID-19 effects. Data shows that older adults have been the most vulnerable to COVID-19, with high hospitalization and death rates. Despite evidence of the vaccine's effectiveness, vaccination rates remain low, and public confidence has decreased. Health leaders stress the importance of clear recommendations and encourage doctors to advise their patients to get vaccinated.