Guinea-Bissau halts US-backed hepatitis B vaccine trial amid ethical and sovereignty concerns
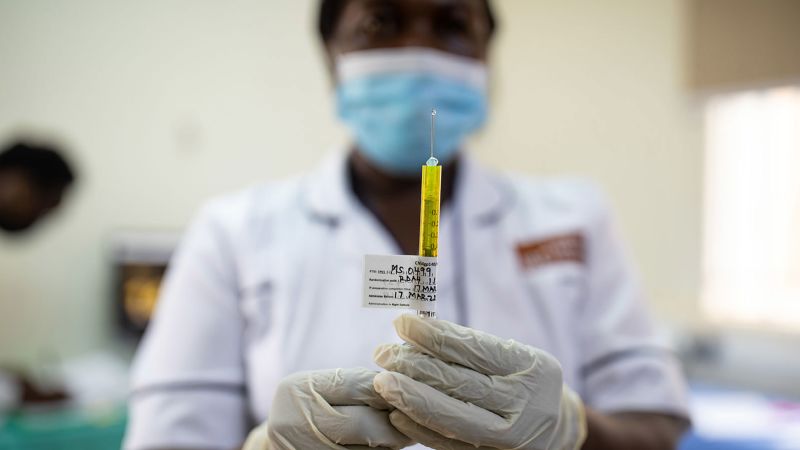
Guinea-Bissau suspended a US-funded, Denmark-led hepatitis B vaccine trial after ethics-review gaps and concerns about consent, with the health ministry citing sovereignty as the deciding factor. The Africa CDC will review the protocol alongside invited US and Danish officials, amid criticism from some experts and a political shift following a coup. The trial design—with 7,000 infants vaccinated at birth and 7,000 withheld for comparison—raised ethical alarms, contrasting with WHO guidance that recommends newborn vaccination within 24 hours and Guinea-Bissau’s current six-week schedule; the outcome will hinge on the ministry’s decision.