High-Dose IV Vitamin C Reemerges as a Potential Cancer Therapy
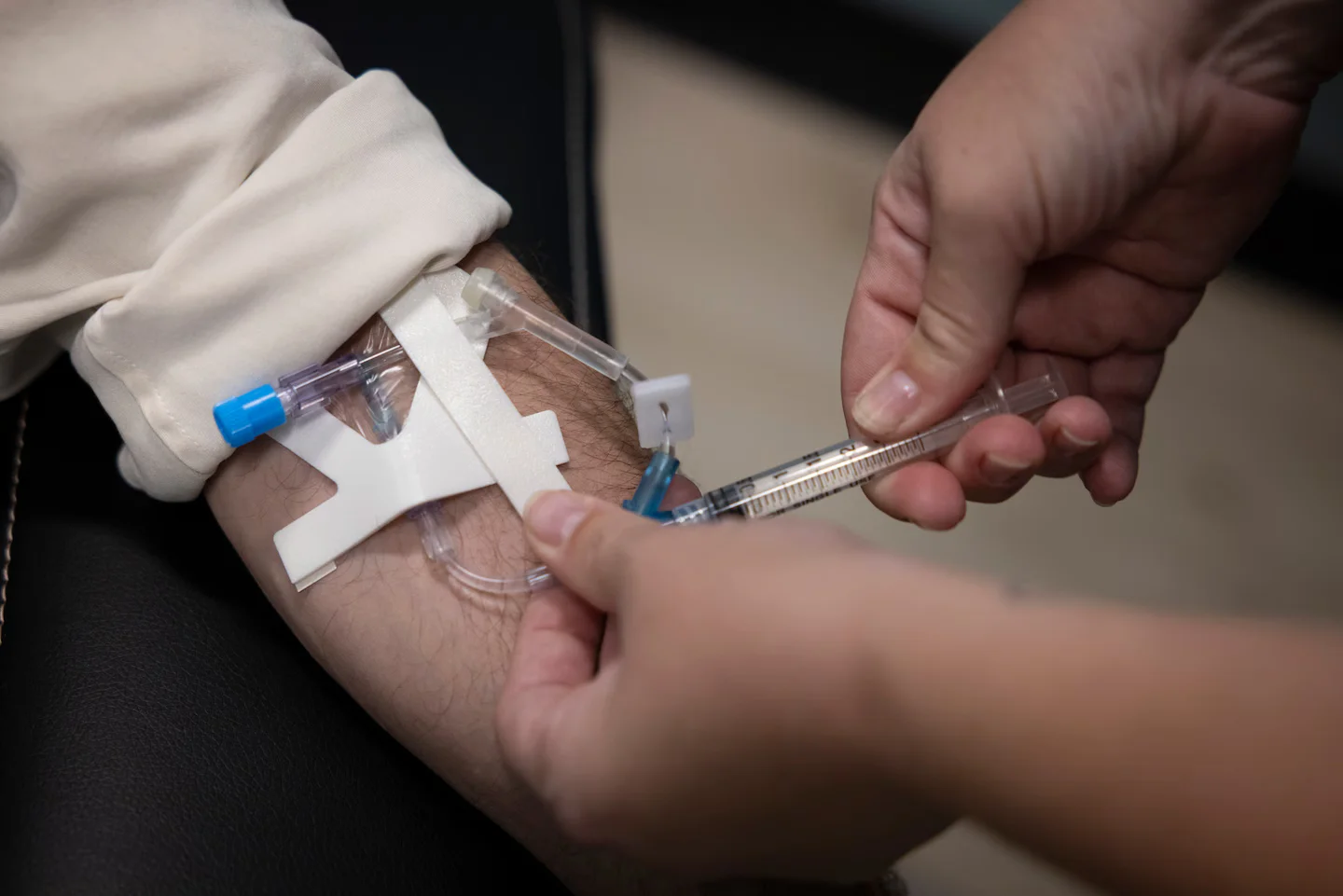
Very high-dose vitamin C given intravenously can reach blood levels far beyond what tablets achieve, where it may generate hydrogen peroxide that selectively damages cancer cells while sparing normal cells; early trials show mixed results—potential improvements in quality of life and some signals of benefit alongside standard treatments, but no definitive survival advantage yet, making it experimental and appropriate only within clinical trials or supervised settings, not as a general cancer cure.