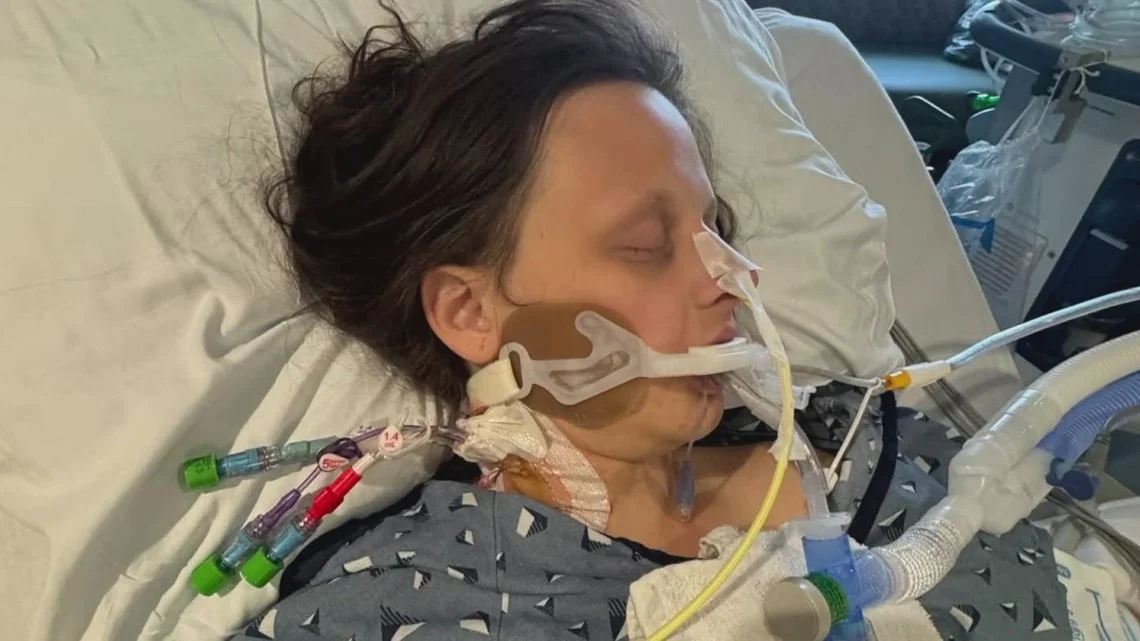
18-Year Odyssey Ends with Rare Jordan Syndrome Diagnosis
After 18 years of seizures, developmental delays and a string of diagnoses that never fit, Lucia Adarve was diagnosed with PPP2R5D-associated neurodevelopmental disorder (Jordan syndrome) via whole-genome sequencing at the Cleveland Clinic Undiagnosed Disease Clinic. The breakthrough provides a clear management plan, reduces seizures, connects the family with support and clinical trials, and gives Lucia a path toward greater independence, ending a long diagnostic odyssey fueled by persistent advocacy from her mother.