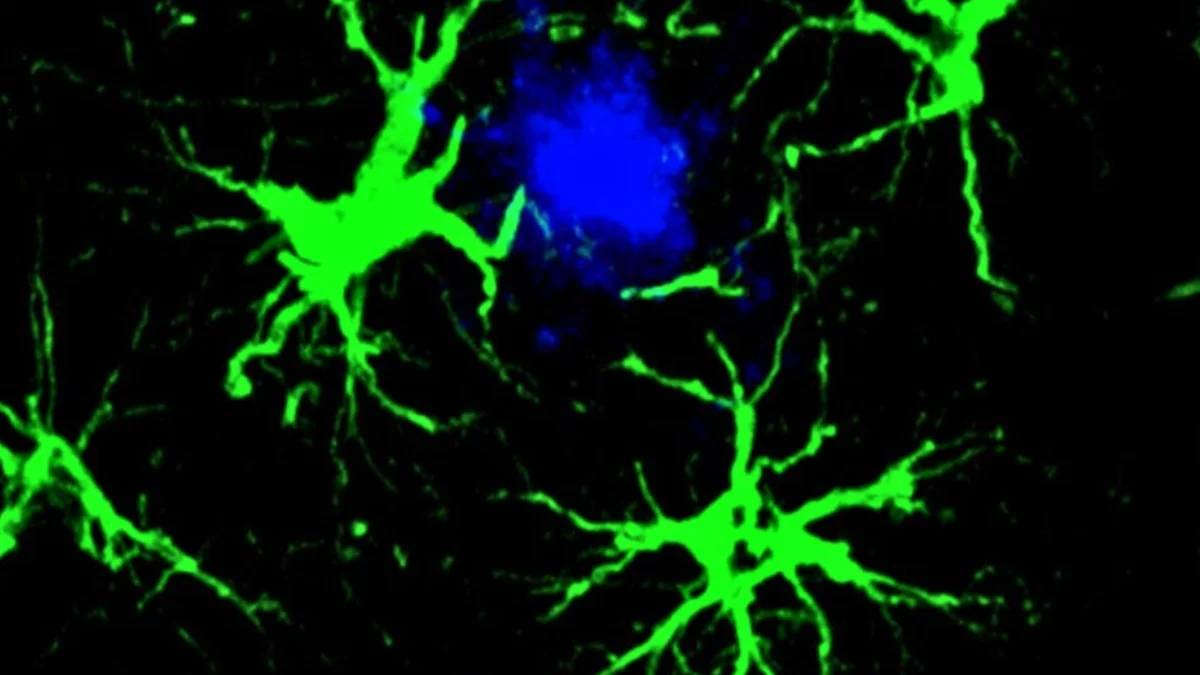
Nanoparticles Restore Brain Cleanup, Reversing Alzheimer’s Signs in Mice
Researchers used supramolecular nanoparticles that act as drugs to repair the brain's blood-brain barrier and restart its waste-clearance system. In mice with high amyloid-β, three injections reduced brain Aβ by 50–60% within an hour, with months-long vascular and cognitive improvements; the approach leverages the LRP1 transport system to reset clearance. While promising, the work is in animal studies and human trials remain years away.